Effective CAPA process explained
Table of contents

When organizations set out to create a set of processes for conducting their work, the focus is on getting things done to meet their customers’ requirements, reduce customer complaints, and ensure that the process is efficient and cost-effective.
At the start of a new process, things usually work well. Still, in time, circumstances change, such as new types of products or services being added, international customers being added, and new staff being recruited. Process compliance is compromised when the process no longer aligns with these changing circumstances, and then problems begin to creep in. These may include orders not being processed correctly or goods and services not being shipped correctly for specific customers.
When this happens, it’s referred to as a deviation from the process or non-conformance. We can document, track, and fix these problems with a quality management or risk management methodology called corrective and preventive action (CAPA). So what is the meaning of corrective action? How can you write a corrective action plan? And what are examples of corrective action or types of corrective actions? This article will answer these questions, and more.
Looking for a CAPA software solution?
What is corrective action?
Corrective action is a systematic process used to identify and address problems or non-conformities in various industries, including manufacturing, healthcare, and finance. It aims to mitigate undesirable results triggered by a task, process, or behavior, and to rectify the process, function, or behavior that adds to the undesirable effects. Corrective action is a crucial aspect of quality management systems, such as ISO 9001, and it’s commonly required in other standards, such as good manufacturing practice (GMP) and hazard analysis and critical control points (HACCP).
In essence, corrective action involves recognizing a problem, determining its root cause, and implementing measures to prevent its recurrence. This proactive approach not only helps in resolving current issues but also in enhancing overall operational efficiency and customer satisfaction. By systematically addressing non-conformities, organizations can ensure compliance with regulatory requirements and maintain high standards of quality management.
Corrective action process
The corrective action process is a series of steps taken to identify, analyze, and resolve problems or non-conformities. It involves gathering data, identifying the root cause of the problem, determining the appropriate corrective action, implementing the solution, and verifying its effectiveness. The corrective action process is an essential part of quality management systems, as it helps to ensure compliance with regulatory requirements, improve business operations, and enhance overall customer satisfaction.
The process typically begins with the identification of a problem or non-conformity, followed by a thorough investigation to determine its root cause. Once the root cause is identified, an appropriate corrective action is planned and implemented. This may involve changes to processes, training for employees, or modifications to equipment. Finally, the effectiveness of the corrective action is verified to ensure that the problem has been resolved and won’t recur. By following this structured approach, organizations can systematically address issues and continuously improve their operations.
Example of a corrective action process
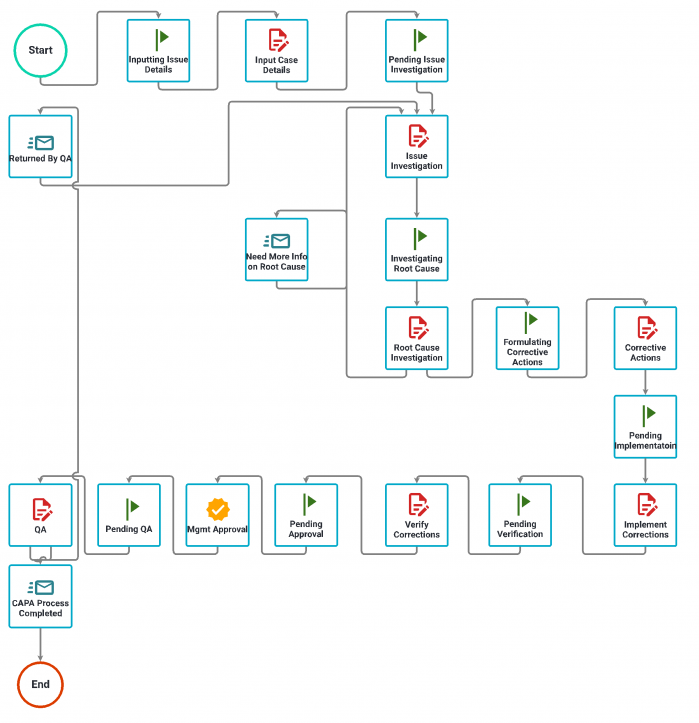
This is a CAPA process built with Nutrient Workflow Automation Platform. The process begins with reporting an issue with details and documentation, and it continues through the investigation and corrections, including review and approval.
Automated CAPA process example
CAPA use cases: Types of corrective actions
There are numerous examples of corrective actions in various industrial and business applications. These range from:
- Major incidents — In global disasters such as the Deepwater Horizon incident in 2010, 11 workers died and 17 were injured, and 60,000 barrels of oil spilled into the sea. The identified cause was the malfunctioning of heavy blades some 20,000 feet underwater. A CAP would have been applied to this and all such significant incidents. In this case, the corrective actions have led to global changes in design and maintenance processes for similar oil and gas installations.
- Deviations from company policy — In HR terms, when employees break a company policy or procedure, they may be reprimanded after such an action. A good example is an employee taking time off work without any notice. In some instances, the deviation may call for a policy change.
- Calibration of tools — In the manufacturing industry, instruments are regularly recalibrated to ensure they measure within set tolerance bands.
- Software fixes — Software applications where the data or the program logic provides erroneous output. Action is taken to identify and remedy the problem.
- Human error or lack of training — Incorrect input to business applications due to human error. This may be a genuine mistake or lack of proper training. Action is taken to provide additional training.
- Equipment/software controls — Triggers and alerts on equipment or built into software when a parameter falls outside a tolerance level. A good example is when an alarm goes off on equipment because the temperature exceeds preset limits.
- Non-adherence to a process/procedure — This occurs when the set process or procedure isn’t followed when completing a task. How often have we assembled furniture from a flat pack and found ourselves so frustrated because we didn’t carefully follow the instructions?
Want to see a CAPA process in action? Check out the automated process(opens in a new tab).
Corrective action process definition
Corrective action is realizing and defining a problem, determining its cause, and taking appropriate measures to prevent it from happening again.
The corrective action process (CAP) then helps to document, create, implement, and verify the actions taken. You’ll often see CAP used alongside the broader term CAPA (corrective and preventive action), the latter of which adds a preventive component aimed at stopping problems before they occur.
A benefit of corrective action: Improving compliance across the organization
We apply “corrective action thinking” in our everyday lives but don’t formally consider it that way. A good example is when we find roadwork on a daily commute, and after experiencing the frustration of the first journey, we find an alternative route. We’ve identified a problem and found a solution that meets our needs.
The difference in an organization is that the problem doesn’t just affect one individual. The consequences of not addressing the issue may be more significant and costly to the organization, hence the need for a formal process. The CAP’s output is an action plan identifying the problem, the root cause, and the resolution.
The concept of corrective action grew out of customer audits of suppliers. Problems, also known as non-conformities or deficiencies, would require corrective actions. The manufacturing industry has long provided suppliers with formats for problem-solving. This same approach has been adopted more broadly into the business process area and incorporated explicitly into the BPM methodology.
We shouldn’t forget that the CAP is applied when a deviation from the process has already occurred. You may ask why the process was set up with known problems in the first place. When developing or reviewing procedures, the objective is to consider all the potential scenarios for possible deviations and incorporate them into the process. However, given the pace of change, it’s a given that change in business circumstances will lead to deviance from the established process.
Best practices for managing noncompliance
When it comes to managing noncompliance in an organization, there are several best practices that can help ensure everyone is held accountable and that the organization operates in an ethical and responsible manner.
- It’s crucial to establish clear policies and procedures that outline employee expectations and consequences for noncompliance. These policies should be communicated clearly and regularly, and employees should receive regular training.
- Creating a culture of transparency fosters accountability. Employees should feel comfortable reporting potential violations without fear of retaliation. There should be accessible channels for reporting and investigating noncompliance.
- It’s important to have a system for monitoring and auditing compliance. This includes regular reviews of financial records, internal controls, and key areas to identify potential issues before they become major problems.
- Having a process in place for responding to noncompliance is crucial. This includes conducting thorough investigations, taking appropriate disciplinary action, and implementing corrective measures to prevent future issues.
Overall, managing noncompliance requires a proactive approach that focuses on prevention, detection, and response. By establishing clear policies and procedures, fostering a culture of transparency and accountability, and implementing effective monitoring and auditing processes, organizations can minimize their risk of noncompliance and ensure they operate with integrity and ethics at all times.
Problem-solving and root cause analysis
Regarding problem-solving and the CAP, a few other techniques are used in various circumstances or specific industries. These techniques include:
- PDCA — Plan, Do, Check, Act. This is an iterative management review method that controls and continuously improves processes and products.
- DMAIC — Define, Measure, Analyze, Improve, and Control. This is a data-driven improvement cycle that’s used for improving, optimizing, and stabilizing business processes and designs.
- 8D — This is a problem-solving methodology that’s designed to find a problem’s root cause based on the ISO 9001 technique.
These techniques complement the corrective action process. Learn more about root cause analysis.
How to write a corrective action plan
There are several variants for applying corrective actions to problem-solving, but they all follow a common approach. The most widely adopted is formalized under the ISO 9001 quality management system, outlined below.
ISO 9001 based — Seven-step corrective action process
- Define the problem — Describe the problem. Make sure the problem is, in fact, a real problem and not a perceived problem. Explain the who, what, when, where, and why. Describe the expected outcome and how the issue can be resolved.
- Define the scope — Make sure you understand how big the problem to be addressed is. Give context to the size of the problem. Is it happening every day? Does it happen only for a particular product, transaction, or customer?
- Containment actions — Make a short-term fix to stop the problem while searching for the ultimate cause and fix. This is about immediate checks or stop-gap measures that will catch the issue again if it recurs while you’re finding the root cause.
- Identify the root cause — Identifying the root cause isn’t always easy. There are specific root cause analysis techniques available to assist with this. The objective is to find the underlying cause, and not just the perceived problem. It’s like a doctor’s diagnosis when the patient complains of tummy pain, but the doctor diagnoses it as something specific, such as diverticulitis.
- Plan a corrective action — Create measurable, achievable solutions with realistic deadlines focusing on the root cause. It needs to include the necessary steps to eliminate the root cause. Depending on the size of the problem, it may require cost and return on investment analysis, with formal management approval to proceed.
- Implement the corrective action — Implement your actions and manage those actions to completion. It could be as simple as making changes to the software or changes in the database. It could be an investment in new equipment in a manufacturing environment because the old equipment can no longer meet tolerance requirements.
- Follow up to make sure the plan worked — Document and close out the process with a debrief to determine what was done and inform the team of the changes. Give sufficient time to ensure that the problem doesn’t manifest again. Capture any lessons learned. Finally, ensure record keeping has been done at an adequate level of detail.
As highlighted, some variants of these steps (refinements to suit specific industries or organizations) exist. The steps may be a simple list or built into sophisticated software solutions. Like all tools, the value is in using them effectively. Simple tools in a craftsman’s hand are better than complex software-based solutions that aren’t fully understood.
If your organization isn’t using this methodology, then guidelines and checklists can be tailored to a company-specific format for the process when someone makes a corrective action request.
Systems for automating and tracking corrective actions
Organizations typically use one of the following systems to manage their corrective action workflows:
- CAPA systems
- Quality management systems (QMS)
- Workflow automation systems
Summary
A corrective action process is a quality management methodology that allows for formal documentation and assessment of industry and business problems.
We recognize that the corrective action process provides a formal, documented means of problem-solving. However, we should remember that solid, lasting corrective actions involve more than filling out a template. They require a disciplined, practical application of the CAPA process.
Finally, let us remember that this methodology helps, but in some circumstances, it may be costly after an event has occurred. Think of the major incidents, other safety applications, and the potential of something going wrong. So, when developing solutions, whether equipment or process-related, we should endeavor to apply a preventive approach (get it right the first time) and avoid using the corrective actions process.
Are you looking to improve and automate your CAPA process?
Nutrient Workflow is a robust process automation platform that enables you to design and deploy your own custom corrective action process, including:
- Incident reporting
- Assigned tasks
- Alerts, reminders, and escalations
- Process tracking
- KPIs and reporting
- Full safety audit trail
Schedule a demo to see a live, automated process and explore what the platform can do.
FAQ
Corrective action addresses existing issues by identifying and solving problems, while preventive action aims to prevent potential future issues.
Automating corrective actions can be achieved through workflow management platforms that help identify, implement, and verify actions systematically.
Root cause analysis helps identify the underlying issue of a problem, ensuring the corrective action addresses the cause rather than just the symptoms.
Yes. Corrective actions are commonly used in software development to fix bugs, improve performance, and ensure software aligns with user requirements.
Compliance ensures that corrective actions are carried out in line with industry standards and regulations, helping organizations maintain operational efficiency and quality.